Electrophysiological Evaluation Of Adolescents Presenting With Chloroquine Toxicity Due To Persistent Malaria Infections
E Anyanwu, I Kanu, J Nnadozie
Citation
E Anyanwu, I Kanu, J Nnadozie. Electrophysiological Evaluation Of Adolescents Presenting With Chloroquine Toxicity Due To Persistent Malaria Infections. The Internet Journal of Neurology. 2005 Volume 5 Number 2.
Abstract
Most drugs used in the treatment of malaria produce phototoxic side effects in both the skin and the eye. These effects are evaluated electrophysiologiclly. The EEG examination was abnormal in 7 out of 10 of the patients compared to the controls with only 1 in 10 with episodes of bi-hemispheric sharp activity. It was later found that this particular control subject has had seizures of unknown etiology in the past. There was fronto-temporal theta wave activity that seemed to indicate diffuse changes characteristic of metabolic encephalopathies. There was scattered and slowing of the alpha rhythm with subsequent appearance of theta and delta activity. The P100 latency was markedly prolonged with mainly the left eye stimulation at an avrage of 143.62 msec and the amplitude of response decreased at 1.26 microvolts. Overall results provided evidence of left optic nerve dysfunction in chloroquine retinopathy.
Introduction
When patients are attacked by malaria (disease caused by plasmodia and transmitted by anopheles mosquito in the tropics) or have rheumatic disorders, they are generally treated with quinine or its derivatives. Almost all patients with hydroxychloroquine (HCQ) retinopathy complain of altered central vision as their first symptom and a normal optic fundus does not exclude the diagnosis HCQ retinopathy (Bienfang et al., 2000). Chloroquine retinopathy (CR) is a major complication of long-term malaria prophylaxis (LTMP) causing permanent visual dysfunction and occasionally blindness. The risk of retinopathy in subjects receiving LTMP is limited to a cumulative dose that does not exceed 140 g. CR that occurred after 8 years of malaria prophylaxis with chloroquine at a cumulative dose of 125 g. Because a threshold dose of chloroquine for retinal toxicity has not been established, careful, ongoing screening is required, especially as the cumulative dose increases (Bertagnolio, et al., 2001). Although, very few patients with mild chloroquine retinopathy may show normal visual acuity, visual fields and full-field electroretinogram, however, retinal dysfunction may be indicated by color vision disturbances. Those with moderate chloroquine retinopathy with normal visual acuity, visual fields and dark-adapted full-field electroretinogram, light-adapted and flicker full-field electroretinogram responses are, however, borderline and color vision was abnormal. While those with severe chloroquine retinopathy show reduced visual acuity, visual field and color vision defects, and a reduced full-field electroretinogram and if color vision defects are detected in patients at risk of developing chloroquine retinopathy, additional testing is indicated to rule out toxicity (Vu et al., 1999; Kellner et al., 2000).
Cutaneous and ocular effects that may be caused by light include changes in skin pigmentation around the ocular adnexia, corneal opacity, cataract formation and other visual disturbances including irreversible retinal damage (retinopathy) leading to blindness (Motten et al., 1999).
The most effective quinine derivative regarded as the drug of choice is choroquine. In treating malaria, chloroquine is used for prevention, treatment of symptomatic attacks of malaria. It is also used for the eradication of plasmodia (radical cure). Other derivatives, which equally are good but not as effective as chloroquine, are amodiaquine, mefloquine and primaquine. Low dose of chloroquine are sometimes given to patients during the symptomatic stage thereby relieving the episodes of fever and chills. However, these medicines do not completely destroy all the malarial parasites remaining in the liver. Hence the attack may re-occur some months or years later. In order to avoid this from happening, patients are usually given the full doses of chloroquine or other quinine derivatives. Such full doses are known to result to serious ocular consequences. Besides the initial manifestations of giddiness, noises in the ear and initial disturbances in vision and hearing, prolonged chloroquine ingestion leads to severe eye damage called chloroquine retinopathy.
Chloroquine Retinopathy
Chloroquine retinopathy is a pigmentary retinopathy which consists of perifoveal change in granularity of RPE, bull's eye macular lesion which shows abnormal electro-oculogram (EOG) or electroretinogram (ERG) but normal dark adaptation. It is prevalent among the peoples of Africa (south of the Sahara), Indian subcontinent, South and Central America, China, Maldives and Mauritius where malaria is endemic and where chloroquine is the most effective therapeutic agent. It may also occur in people from other nations who have lived or are still living in the malarial endemic regions. It could also occur among travelers to these endemic regions. In this case they contract the malaria and are usually treated with chloroquine. It must be stated that it is most a prolonged chloroquine treatment either as a curative measure or as prophylaxis that causes the most harm to the eye. Chloroquine retinopathy in addition, occurs in patients who take chloroquine and its related drugs for rheumatoid arthritis or systemic lupus erythematosus. The worst thing about treating patients with rheumatic disorders with chloroquine is that the side effects of chloroquine retinopathy may occur long before any beneficial effects can be achieved.
Differential Diagnosis
Choroquine retinopathy can effectively be differentiated from other types of cone degeneration which show bull's eye lesion in that all other cone degeneration are autosomal dominant with the following symptoms occurring in the first two decades of life: severely reduced acuity, color vision defects, photophobia and nystagmus. Most of these symptoms, if they ever occur do so in a lesser degree. However, photophobia persists throughout life depending on the age-related effects and prolonged chloroquine ingestion. Dark adaptation is generally unaffected by chloroquine ingestion thus, differentiating it from retinitis pigmentosa. It is advisable that the accurate differentiation of chloroquine retinopathy should be based on the previous knowledge of the patient's history of chloroquine ingestion.
Clinical Findings and Complications
The initial changes in the paracentral retinal function with central visual acuity are subtle but manifest itself after a short while. Gradually a bull's eye pattern of clumping and thinning of the retinal pigment in the macular area with an annular scotoma within 2 to 3 degrees from fixation. This condition may lead to progressive central vision loss and the peripheral field changes. Although, dark adaptation remains functionally good and the rod limb of the dark adaptation curve is markedly unaffected, however, the drug appears to selectively destroy cone function. Such findings are cumulative more especially if the patient has ingested chloroquine over periods of months or years. This appears to be caused by very tight binding of the drug to pigment within the pigment epithelial cells of retina. Usually, a marked depressed EOG or an enlarged A-wave or reduced B-wave of ERG is observed. The visual acuity is markedly reduced (20/200 or worse) in later stages. Color vision is also affected. For an accurate detection of the bull's eye, and to observe the defects in the cone limb of the dark adaptation curve, small test lights confined to the central macular area of the visual field is used. Complications of chloroquine retinopathy may very according to the periods of prolonged chloroquine ingestion. Corneal deposits and pronounced vascularization may be observed. Visual acuity defects may be accompanied by an appearance of colored halos around lights. Age-related myopia increase in severity with prolonged chloroquine ingestion.
Etiology and Pathogenesis
Choroquine retinopathy is caused by the ingestion of chloroquine or its related sulfates and disulphates of over 250 mg per day to a total exceeding 500 mg. The onset may vary according to the amount of in-take and upon individual eye condition; it may also occur one to three years after the medication is first taken. Chloroquine is known to inhibit DNA and RNA synthesis and has an enormous binding capacity to pigmented tissues of the yes. Ganglion cell changes are seen first, followed by photoreceptor degeneration. Occasionally, upon cessation of drug therapy, the lost vision is recovered. Usually, the patient remains stable after medication is discontinued. In some cases, the disease has progressed even after cessation of chloroquine ingestion.
Materials and Methods
The study population
Ten young adults aged between 14 and 19 years old, 6 (60%) female and 4 (40%) male, and 10 sex and age-matched controls made up the study population. A majority of the adolescents came from areas that were endemic to malarial infections. Less than 30 % of the adolescents were down with malaria at least once a month. The controls on the other hand had rarely had malaria every year and did not take malarial drug within six months before the study. They all had no known history of any neurological disorders.
Electrophysiological tests
Carefully designed neurological questionnaires were first administered with the cooperation of the children's parents. Then, all the patients and controls underwent clinical neurophysiological tests including EEG, visual evoked potential (VEP), and nerve conduction velocity using Biologic CEEG Graph 4 Model 810 Version 5.71 with Biologic EP Explorer. All tests were carried out according to the standard methods. The EEG examination included hyperventilation and intermittent photic stimulation (IPS) using Biologic PS2 photo stimulator at intensity 1363 cd/m2. International electrode placement 10-20 system was used. The photostimulator was positioned at a distance of 18 inches (45.72 cm) from subjects' eyes at a visual angle of 25 degrees and was given first with the eyes open for 5 seconds, then 5-second eye closure, and another 5 seconds with the eyes closed. Both the patients and the controls were asked to sit at a distance of 170 cm from the stimulus source with their eyes fixated on the red spot at the center of the monitor. Nerve conduction velocity was carried out using TECA Synergy Multimedia EMG with multi-sync color SVGA monitor and Delux stimulator probe (TECAsynergy, Oxford Instruments Medical Inc). VEP test was performed with corrective lenses when needed. For the reliability and reproducibility of tests, repeats were performed.
Data analysis
Data generated from the questionnaire were analyzed using power formula analysis (Amler et al., 1996) to calculate the number required to detect a difference between the mean score of the patients (abnormal) and controls (normal) responses. Values from power analysis formula for n to detect a 20% difference was 0.05 for abnormal; 0.05 for normal. Power (P) was 95% and the constant (K) was 3.29. Neurological response differences in mean change were assessed using an analysis of variance (ANOVA) model. Paired t-test comparisons between outcomes in each were conducted using least squares means from the above model. Ninety-five percent confidence intervals for effect-exposure differences were computed on the basis of the least squares means from the ANOVA model. For binary measures, such as the percentage of neurological abnormalities, comparisons were conducted using Chi-square test. Unadjusted P values are reported. To assess symptom severity at endpoint relative to an unaffected population, we analyzed scores on the primary outcome measure as t scores. All statistical analyses were performed using StatiBot 2000-2002 software.
Results
Most drugs used in the treatment of malaria produce phototoxic side effects in both the skin and the eye. Pathologic findings in chloroquine retinopathy are shown in Table 1.
EEG examination
The EEG examination was abnormal in 7 out of 10 of the patients compared to the controls with only 1 in 10 with episodes of bi-hemispheric sharp activity. It was later found that this particular control subject has had seizures of unknown etiology in the past. There was fronto-temporal theta wave activity that seemed to indicate diffuse changes characteristic of metabolic encephalopathies. There was scattered and slowing of the alpha rhythm with subsequent appearance of theta and delta activity. This wave activity was enhanced by hyperventilation. There was certainly undefined triphasic wave activity in a majority of the patients that consisted of a minor positive potential preceded and followed by another smaller negative waves. In 3 out of 10 patients in particular, there was highly marked 1-3 Hz delta activity that was asymmetrical in the right hemisphere of the brain. Sharp synchronous bilateral theta activity of 4 Hz was also observed and there were no such wave activity in the controls.
Table 1: Findings in Chloroquine Retinopathy
VEP examination
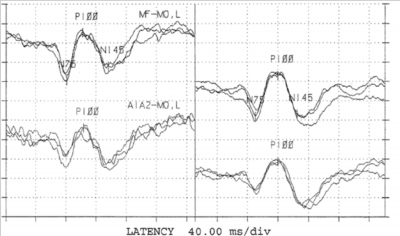
Visual evoked response testing was carried out stimulating each eye independently with pattern shift stimulus. Although, the waveforms were well developed bilaterally in all the patients, the VEP showed clear abnormalities in 4 in 10 of the patients compared to 1 in 10 of the controls. Four in 10 of the patients' P100 amplitudes and latencies were decreased bilaterally. The mean full field pattern reversal for P100 latency was OS 120.17:OD 119.19 milliseconds in the patients compared to OS 93.79:OD 93.63 milliseconds in the controls. The mean P100 amplitude was OS 6.82: 5.19 microvolts compared to OS 21.42:OD 20.18 microvolts for the controls. On hemifield testing there was also a definite prolongation of the latencies bilaterally in all the patients during stimulation. These findings may represent evidence of bilateral optic nerve dysfunction. had visual disturbances with a visual acuity of 20/30 in the right eye.
Figure 2
It was not possible to verify the visual acuity of the left eye of one of the patients probably due to nystagmus in that eye. Although, the waveforms were well developed with right eye stimulation and less developed with left eye stimulation. The P100 latency was markedly prolonged with left eye stimulation at 143.62 msec and the amplitude of response was decreased at 1.26 microvolts. The P100 latency and amplitudes were well within normal limits with the right eye stimulation. This is an evidence of left optic nerve dysfunction.
Discussion
Although relatively rare, retinopathy based on a disturbed metabolism of the retinal pigment epithelium (RPE), with ensuing degeneration of photoreceptors, is a known complication of treatment with the 4-aminoquinolones, chloroquine (CQ) and hydroxychloroquine (HCQ), in autoimmune diseases. The reported frequency of retinopathy, however, is much lower for HCQ than for CQ (less than 0.08% versus 1-2%) (Easterbrook, 1999; Sundelin and Terman, 2002). Risk factors for the development of toxicity are daily dosage related to body weight, total drug dosage and the specific drug used. The daily dosage of chloroquine should not exceed 4 mg/kg lean body weight a day. Exceeding the total dosage of 300 g increases the risk. The daily dosage of hydroxychloroquine should not exceed 6.5 mg/kg lean body weight a day with increased risk when the duration of treatment exceeds 8 years (1330 g for a patient of 70 kg). Regular dilated fundus examination, visual field testing using Amsler grid test and automated central 10-2 perimetry (to a red test object) is advised to detect reversible premaculopathy (Herman et al., 2002)
CRP in the regions of the world where malarial infection is rare, are probably due mainly to over dosage in the treatment of dermatitides of systemic lupus erythematosus and rheumatoid arthritis (Wang et al., 1999; Wei et al., 2001). All cases arose because of failure by physicians to avoid dosing above published safe levels. Five cases developed despite accepted ophthalmologic patterns of screening for toxicity. All cases developed parafoveal retinal pigment epithelial atrophic changes and paracentral scotomas to threshold visual field testing. New cases of hydroxychloroquine and chloroquine toxicity continue to develop in a screening environment. Increased ophthalmologic attention to dosing, awareness of location and nature of early visual field defects, and traditional attention to presence or absence of maculopathy can reduce the incidence of this avoidable condition (Browning, 2002).
Chronic use of chloroquine and hydroxychloroquine in the treatment of rheumatic disease carries a small risk of sight-threatening pigmentary retinopathy. To obtain safety data for its use in pregnancy, we did ophthalmic examinations in 21 children born to women who took these drugs during pregnancy. Average daily maternal doses of the two drugs were 317 mg hydroxychloroquine and 332 mg chloroquine. The mean duration of gestational exposure was 7.2 months. No ophthalmic abnormality was detected in these children. Therapeutic use of these drugs during pregnancy may not pose a significant risk of ocular toxicity to offspring (Klinger et al., 2001).
The frequency of occurrence of retinopathy varies from 0.001 to 40% depending on the criteria used. Most patients received 250 mg of chloroquine per day. The duration of treatment varied from 6 months to 14 years, and the cumulative dose of chloroquine ranged from 26 to 1771 g. Fourteen patients (9%) had only corneal deposition, while 22 (14.2%) developed retinopathy. There were no correlations between corneal deposits or retinopathy and age, sex, duration of treatment, or cumulative dose of chloroquine. Retinopathy is not related to the duration of treatment and cumulative dose of chloroquine. However, retinopathy can be detected as early as 9 months after starting chloroquine therapy (Mackenzie, 1983; Puavilai et al., 1999).
Electrophysiological findings are usually pathologic in patients with chloroquine-induced bull's-eye maculopathy. To avoid maculopathy the daily dosage of chloroquine is estimated not from the actual but from the ideal body weight and should not exceed 3.5 mg/kg/day (Cursiefen et al., 1997). Patients usually complain about blurred vision, with visual acuity from 0.8 to 0.3 (right eye) and 0.4 (left eye), a central scotoma is present and fundus-examination shows a typical bull's-eye maculopathy. In spite of the prominent fundoscopic changes the electrophysiological examination (ERG, EOG and pattern-ERG) may be normal (Cursiefen et al., 1997) probably because of the relative smallness of affected retina. Patients receiving antimalarial therapy, specifically hydroxychloroquine, for different periods show macular dysfunction (Bishara & Matamoros, 1989;Levy et al., 1997).
Conclusions
The electrophysiological evaluation of adolescents presenting with chloroquine toxicity due to persistent malaria infections has been done. The findings are pathologic in the patients with chloroquine-induced bull's-eye maculopathy. Patients receiving antimalarial therapy, specifically advised to have their eyes examined at regular intervals. Routine ophthalmic screening is indicated if the daily dosage of hydrochloroquine is >6.5 mg/kg or who have taken HCQ continuously for > 10 years.