Intra-Thrombus Lytic Therapy May Reduce Complications Of Primary Percutaneous Intervention In Acute Myocardial Infarction And Massive Intracoronary Thrombus Not Treated With Glycoprotein IIb/IIIa Antagonists
T Sakuma, T Tokuyama, T Oka, K Miura, K Ishibashi, T Okada, M Otsuka, M Toyofuku, H Hirao, Y Muraoka, H Ueda, Y Masaoka, Y Hayashi
Keywords
acute myocardial infarction, distal microcirculatory protection, reperfusion, thrombolysis
Citation
T Sakuma, T Tokuyama, T Oka, K Miura, K Ishibashi, T Okada, M Otsuka, M Toyofuku, H Hirao, Y Muraoka, H Ueda, Y Masaoka, Y Hayashi. Intra-Thrombus Lytic Therapy May Reduce Complications Of Primary Percutaneous Intervention In Acute Myocardial Infarction And Massive Intracoronary Thrombus Not Treated With Glycoprotein IIb/IIIa Antagonists. The Internet Journal of Cardiology. 2005 Volume 3 Number 2.
Abstract
Introduction
Although a thrombus burden further enhances the no-reflow phenomenon shortly after coronary recanalization in acute myocardial infarction (AMI) experiments in animals1,2,3, randomized trials in patients with AMI using a distal coronary protective device and thrombus aspiration system have shown disappointing results in terms of effectiveness for salvaging jeopardized myocardium.4 We hypothesized that thrombus could not be retrieved well even after performing repetitive thrombus aspirations during distal coronary protection because the fibrin mesh would not have been resolved.5 Therefore, this study aimed to investigate whether repetitive aspirations of thrombus with fibrinolysis under distal coronary protection is useful for reducing procedural complications during primary percutaneous coronary interventions (PCI), and for preserving left ventricular (LV) function by reducing total amounts of distal thrombo-emboli.
Methods
Patient selection
This retrospective and partially prospective study comprised 44 consecutive patients having their first acute inferior myocardial infarction accompanied with massive static coronary thrombus as detected by coronary angiography. Coronary thrombus was visually assessed by the modified method based on the definition by Muhlestein et.al.6 Definitive thrombus greater than 4-fold of reference coronary diameter was determined as a static massive thrombus. Patients with inferior wall AMI indicated by ST elevation >0.1 mV in inferior leads, significant elevation of CK-MB, echocardiographic evidence of dys-synergy in inferior and/or posterior wall and angiographically proven thrombus in the right coronary artery were included in the study. All patients gave informed written consent for PCI and the present study was approved by our institutional review board. Informed written consent for participating in a prospective part of this study was obtained from corresponding patients. Since several parts of this study were retrospective, we obtained written consent from all patients for permission of use of their data.
Interventional procedures
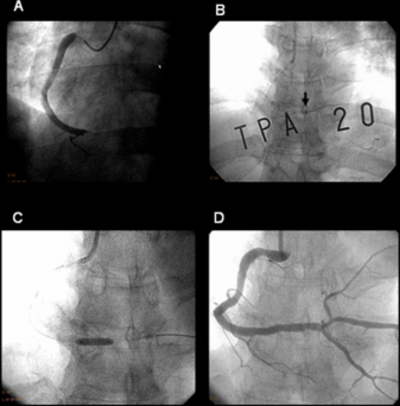
All patients took 200mg oral aspirin and 200mg ticlopidine, then underwent emergent coronary angiography after administration of 50units/kg intravenous heparin sodium as well as 1mg isosorbide dinitrate for each coronary artery. A static massive thrombus on the culprit lesion was confirmed on the second or third segment of the right coronary artery. After the intravenous administration of an additional 100units/kg heparin sodium, wires were inserted into the main right coronary artery as well as the distal major side brunch for preventing distal coronary thrombo-emboli. PCI using a conventional balloon and/or bare metallic stents was performed within 6 hours from the onset of symptoms, Thirty two of 44 patients (Group 1) underwent PCI using the Guardwire Plus System® (GPS, Medtronic Japan, Kawasaki, Japan);4 in these patients coronary protective balloon was inflated at the distal portion of the third segment where the collateral artery could be detected on the coronary angiography. Then, thrombectomy was conducted prior to coronary stenting using the Export® aspiration catheter that was part of the GPS. Repeated aspiration of thrombus as well as the plaque debris was carried out before and shortly after coronary stenting (Figures1 and 2).
Five of the 44 patients (Group 2) underwent intra-thrombus lytic therapy (ITLT) alone using a 200, 000 IU mutant tissue plasminogen activator (monteplase, Eisai,Tokyo Japan) 7 on the site of the coronary thrombus through a FC-catheter (Tokai Medical Products, Aichi, Japan), which is a monorail type catheter with distal side holes for drug infusion. Another 7 patients (Group 3) underwent ITLT with a GPS; in these repeated thrombectomy was carried out during distal coronary occlusion shortly after ITLT as well as shortly before and after coronary stenting (Figures 1 and 2). Twenty three patients in group 1 were retrospectively involved in this study. Other 9 patients in group1 and all patients in group 2 as well as group3 were enrolled prospectively.
Quantitative analysis
TIMI grade flow,8 myocardial blush grade (i.e. tissue myocardial perfusion grade),9 ST segment re-elevation (>1.3 times of baseline ST segment elevation),10 and ST segment resolution (>50% of baseline ST segment elevation)11 were assessed using previously reported definitions. Quantitative coronary and LV functional analyses were performed using methods similar to those described elsewhere.12,13,14,15 LV function was re-assessed using left ventriculography at 6 months after primary PCI.
Statistical analysis
Results are expressed as mean values ±1 standard deviations (SD) or as proportions. Differences among 3 groups were analyzed by 1-way analysis of variance and Scheffe's post hoc test in continuous variables or Kruskal-Wallis test with Tukey's post hoc test (non-parametric multiple comparative test) in discrete variables or chi-square test with Bonferroni's post hoc test in the proportions. The relation between frequencies of procedural complications and baseline characteristics was examined by forward stepwise multiple linear regression analysis, with the F value equal to 4.0. Statistical significance was defined as a p value .0.05 (2-sided).
Results
Patients' clinical and angiographic characteristics are documented in table 1. Cases which ultimately developed Q-waves after primary PCI were defined as Q-wave myocardial infarction. Complications during PCI and any additional use of medicinal treatments during PCI are shown in table 2.
Eighteen patients (56%) in group 1 were documented as having significant complications (distal coronary emboli (n=7), hypotension (systolic arterial blood pressure < 80 mmHg) (n=4), malignant arrhythmia (n=8), side branch occlusion (n=3), and ST-re-elevation with worsened chest pain (n=9) Two of these 7 patients with distal coronary emboli still had insufficient coronary flow even after adjunctive medicinal treatment. In group 2, all patients showed complications. (transient distal coronary emboli (n=2), prolonged hypotension with bradycardia (n=1), and worsened chest pain with ST-re-elevation (n=3). However, no patients in group 3 showed any significant complication except for transient distal coronary embolisation that had been resolved after additional treatments(n=2). A typical example is demonstrated in Figure 3.
Figure 5
TIMI grade flow at the final angiography,8 ST-segment resolution,11LV ejection fraction and LV end-diastolic volume indices were significantly different among the 3 groups. Only the strategy of PCI was a significant determinant (F value = 11.0, p=0.0008) for frequencies of complications during PCI when we set the number of complications in individual patients as a dependent variable; and age, gender, Killip class, infarct related arterial segment, extension of diseased vessels, LV ejection fraction on arrival, coronary occlusion time, collateral circulations16 before PCI and strategies of PCI as independent variables by a stepwise method on multiple linear regression analysis. However, we could not determine any significant variables for our study patients when TIMI grade flow at the final angiography, or ST-segment resolution, or LV ejection fraction or LV end-diastolic volume indices were set as dependent variables in similar multivariate analyses.
Discussion
In this study we have elucidated several advantages of simultaneously using both GPS and ITLT in patients with inferior AMI complicated by massive thrombus. In general, the risk area in inferior AMI is smaller than that in anterior AMI.13Hence, final infarct size tends to be smaller. However, primary PCI using metallic stents occasionally causes no or low-reflow phenomena14 when carried out without any distal coronary protection. Microembolisation into distal coronary territory as well as microvascular damage due to ischemia lead to no-reflow phenomena.1 The no-reflow zone within the initial risk area progressively increases in size for several minutes, or even hours, shortly after coronary recanalization.2?Leukocytes, platelets, and adhesion molecules also contribute to these processes.3 Therefore, special attention needs to be paid to the PCI strategy immediately before and shortly after coronary recanalization. In cases where GPS is not used, unresolved massive thrombus that slip down into distal coronary region might cause distal coronary embolization.1 As well, several massive thrombus can not be fully resolved due to the extensive fibrin-mesh formation.5 Our proposed PCI strategies might affect a lesser level of injured microvascular integrity within the initial risk area. Although these procedures can appear slightly complicated and prolong the required time, these strategies should be able to be safely accomplished in most cases, except for patients who have massive thrombus in an ostial lesion of the right coronary artery.
Study limitations
Several limitations underlay this study. This was a highly selective, small, non-randomized study without a definitive control group. Our findings need to be confirmed in a prospective large cohort study. We could not set a definitive controlled group for ethical reasons, i.e. a group with PCI but without any additional treatment. However, our three groups indicated similar baseline characteristics, and multivariate analysis validated part of our hypotheses. In Japan, IIb/IIIa antagonists3 have not yet been made available for general clinical use; the additional minimum use of intra-coronary administration of IIb/IIIa antagonists with tissue plasminogen activator17 might have shown greater effectiveness in reducing procedural complications. We could not objectively measure the amounts of thrombus burden retrieved after completion of aspiration.2 Finally, right ventricular function18 as well as microvascular integrity could not be evaluated in detail.
Conclusions
In spite of the small study cohort, ITLT using an FC-catheter followed by PCI with a GPS may reduce procedural complications during PCI in patients with inferior wall AMI and massive intracoronary thrombus not treated with glycoprotein IIb / IIIa inhibitors. These results may indicate a promising strategy in patients with inferior AMI complicated by massive thrombus.
Correspondence to
Tadamichi Sakuma, MD, FACC, FESC, 3-30 Nakashima-cho, Naka-ku, Hiroshima, Japan, Postal code 730-0812 Telephone: 81-82-243-9191 Fax: 81-82-241-1865 e-mail: tasakum@hotmail.com