Adult Onset Type 1 Diabetes Mellitus Versus Type 2 Diabetes Mellitus: A Case Study
E Ballard
Keywords
diabetes mellitus, ketosis-prone type 2 diabetes mellitus, latent autoimmune diabetes in adults, type 1 diabetes mellitus; type 2 diabetes mellitus
Citation
E Ballard. Adult Onset Type 1 Diabetes Mellitus Versus Type 2 Diabetes Mellitus: A Case Study. The Internet Journal of Advanced Nursing Practice. 2008 Volume 10 Number 1.
Abstract
A 34 year old Caucasian male with symptoms of polyuria, polydipsia, blurry vision and a 40 pound weight loss over a two month period presented to an outpatient specialty clinic. This article describes the evidence-based evaluation and management by the advanced practice nurse (APN) of a patient with newly diagnosed diabetes mellitus. Special emphasis is given to the criteria necessary for differentiation between type1 diabetes mellitus (T1DM) and other types of diabetes, particularly type 2 diabetes mellitus (T2DM) and latent autoimmune diabetes in adults (LADA).
Introduction
Differentiation of type 1 diabetes mellitus (T1DM) from type 2 diabetes mellitus (T2DM) in adults may not always be straightforward. Furthermore, the differentiation of Ketosis-prone T2DM (KP-T2DM) and Latent Autoimmune Diabetes in Adults (LADA) from T1DM is especially difficult at times. Correct differentiation between T1DM and these other types of diabetes is important in developing a treatment plan, in particular, whether or not to initiate insulin. [1] This case study presents an example of criteria useful in determining the correct diagnosis and initiating treatment.
Case Report
History of Present Illness
Two weeks after being diagnosed by his Primary Care Provider (PCP) with T2DM, a 34 year-old white male was referred to the Diabetes Specialty Care Team for education and evaluation. Despite a 40-pound weight loss over the past two months, the PCP diagnosed the patient with T2DM. The patient also reported having vision problems for two months prior to diagnosis of T2DM. As a result of his history of Pigmentation Disbursement Syndrome, the patient initially made an appointment with an ophthalmologist. He believed that his eye disorder was the cause of his vision changes. However, after several appointments the ophthalmologist determined that the symptoms were unrelated to his eye condition and suggested he see his PCP for further evaluation. The patient also reported symptoms of polyuria, polydipsia, fatigue and frequent headaches for several weeks in addition to blurry vision and the 40-pound weight loss. Results of laboratory tests ordered by the PCP at the initial appointment are listed in Table 1.
A lipid panel and dipstick urinalysis was not available for review. Despite these lab results indicating early metabolic acidosis, the patient denied having any symptoms of diabetic ketoacidosis (DKA); a potential risk. The symptoms of DKA include lethargy, confusion, extreme fatigue, abdominal pain, vomiting, extreme thirst, or dry mucous membranes. [2]
The fasting blood glucose during the two weeks since the patient’s initial diagnosis ranged from 227 - 455 mg/dL. The post-prandial glucose and bedtime glucose readings ranged from 240 - 595 mg/dL. At the initial appointment with the PCP, the patient had been placed on metformin (Glucophage ® ) 500 mg twice daily and pioglitazone (Actos ® ) 15 mg once daily. However, he continued to experience polydipsia, polyuria, headaches, and blurry vision.
Past Medical History
The patient’s past medical history was significant for mild-intermittent asthma since childhood, seasonal allergic rhinitis, diverticulosis, and pigment dispersion syndrome of the eye. This chronic problem is the result of a loss of pigment from the neuroepithelial posterior surface of the iris, which is spread throughout various structures of both anterior and posterior chambers. The pigment is carried forward and deposited along the routes of aqueous flow, creating decreased outflow of the trabecular meshwork. The decreased outflow can potentially result in glaucoma. [34] The patient believed that his blurry vision was related to his chronic eye problem, and initially sought care from his ophthalmologist.
Allergies included penicillin. His surgical history was significant for an appendectomy and bilateral inguinal hernia repairs as a child. Current medications included montelukast (Singular ® ) 10 mg p. o. once daily; mometasone (Nasonex ® ) nasal spray 50mcg/spray-2 sprays each nostril once daily; metformin (Glucophage ® ) 500 mg p. o. twice daily (recently started by PCP), and pioglitazone (Actos ® ) 15 mg once daily (recently started).
Family History
Family history was significant for hypertension (HTN) and elevated cholesterol. There was no family history of T1DM or T2DM diabetes in his family.
Social History
The patient was married and had one three year-old child. His wife was very supportive and accompanied him to his appointments. He denied smoking, alcohol or illicit drug use. He was employed full time and had insurance coverage.
Physical Exam
Physical exam revealed a 34-year-old slightly overweight Caucasian male in no acute distress. Height: 74 inches; Weight: 202 lbs; Body Mass Index (BMI): 25.9 kg/m [2] ; Waist circumference: 44 inches; Temperature: 97.9 F; Pulse: 82 beats/minute; blood pressure (BP): 108/78 mmHg; Respirations: 16/minute. The patient was well developed, well nourished and his appearance was consistent with his stated age. He was dressed appropriately for the weather and was well groomed. Pupils were equal, round and reactive to light and accommodation, with a faint circular line of darker pigment around the outer edges of both irises. Extraocular movements were intact in all six fields without nystagmus. The remainder of the physical exam was unremarkable.
Differential Diagnoses
-
Type 1 Diabetes Mellitus
-
Type 2 Diabetes Mellitus
-
Ketosis-prone Type 2 Diabetes Mellitus (KP-T2DM)
-
Latent Autoimmune Diabetes in Adults (LADA)
-
Diabetic Ketoacidosis (DKA)
-
Hyperglycemic Hyperosmolar Nonketotic State (HHNS)
-
Ketosis with metabolic acidosis
Differentiating between T1DM and T2DM is not always straightforward. The patient presented with the typical symptoms of hyperglycemia, including polyuria, polydipsia, blurry vision, and fatigue. In addition, he had experienced a 40-pound weight loss over the past two months. The in-office laboratory findings at the first appointment to the diabetes specialty care team revealed 4+ ketones in the urine, urine glucose greater than 2000+, and a random fingerstick glucose of 395 mg/dL. Review of the laboratory ordered by the PCP two weeks earlier was significant for early metabolic acidosis, as exhibited by the low C02 (20 mEq/L), in conjunction with extremely high urine ketones, despite a normal anion gap. The reported C02 in the serum refers to the total C02 in the blood, including that contained in bicarbonate. More than 70% of C02 in the blood is in the form of bicarbonate. [5] Causes of non-anion-gap acidosis include ketosis with ketone excretion. [6] Metabolic acidosis is defined as a decrease in bicarbonate. Metabolic acidosis may be caused by an overproduction of keto-acids with a respiratory alkalosis due to the compensatory increase in ventilation, in an effort to decrease C02 and bicarbonate (H2C03) levels. [5]
Polyuria is a result of osmotic diuresis secondary to sustained hyperglycemia. Blurry vision and increased thirst are also a result of this hyperosmolar state. Weight loss is due to the breakdown of fat and muscle stores for the production of energy. [2] According to Gardner and Shoback, even a small amount of insulin can prevent the development of ketosis by inhibiting lipolysis in the adipose stores. Therefore, development of ketonuria is an indication of absolute insulin deficiency. [2] Ketonuria is very common in T1DM, but is rarely seen in T2DM, with the exception of KP-T2DM, where this finding can be seen. [7] The presence of urinary ketones alone should cause the practitioner to consider T1DM as a differential diagnosis, and insulin initiation is warranted.
Acute life-threatening complications of uncontrolled diabetes are hyperglycemia with ketoacidosis or nonketotic hyperosmolar syndrome. [89] Of the two, only diabetic ketoacidosis (DKA) presents with elevated ketone bodies in the blood and urine ketones. In DKA, the lack of insulin stimulates lipolysis in the adipose tissue, and ketogenesis in the liver, resulting in increased free fatty acids and a rise in hydrogen ions leading to metabolic acidosis. [10] The hyperglycemic, hyperosmolar, nonketotic state (HHNS) is characterized by severe hyperglycemia, hyperosmolality, and dehydration without significant ketosis. A partial or relative insulin deficiency leads to decreased glucose utilization by the muscles, and increased hepatic glucose output. The result is a hyperglycemia that leads to glycosuria, osmotic diuresis, and dehydration. The presence of even a small amount of circulating insulin can prevent ketosis by inhibiting lipolysis. The inhibition of lipolysis and subsequent lack of available free fatty acids restricts the rate at which ketones are formed, thereby preventing severe ketosis. If a patient is unable to maintain adequate fluid intake, severe dehydration can occur, leading to renal insufficiency, decreased glucose excretion, and a marked rise in serum glucose and osmolality. Serum osmolality above 320-330 mosm/kg results in water being drawn out of cerebral neurons, resulting in mental obtundation and coma. [2]
Assessment/Impressions
-
T1DM
-
Ketosis with metabolic acidosis.
Plan
Laboratory Tests
-
Complete metabolic profile
-
Lipid profile
-
Urine microalbumin
-
Islet cell autoantibodies (ICAs) included: 1) insulin autoantibodies (IAA), 2) protein
-
tyrosine phosphatase (IA-2A), and 3) glutamic acid decarboxylase – 65 (GAD-65).
-
C-peptide level
Diabetes mellitus is a chronic disorder of the metabolism due either to a deficiency of insulin secretion, a decreased effectiveness of insulin, or both. [2] The etiology is believed to be the result of an interaction between the environment and genetics of susceptible individuals. The fasting plasma glucose is the preferred test for diagnosing diabetes in children and non-pregnant adults. [8] Although the 75 gm oral glucose tolerance test is slightly more accurate, the inconvenience and time required make this diagnostic test less desirable. The provisional diagnosis of diabetes requires one of the following criteria: 1) fasting plasma glucose greater than 126 mg/dL after an eight hour fast; OR 2) symptoms of hyperglycemia (polyuria, polydipsia, and unexplained weight loss) and a random plasma glucose of greater than 200mg/dL; OR 3) a 75 gm 2-hour oral glucose tolerance test (OGTT) of greater than 200 mg/Dl (Table 2). The diagnosis must be confirmed on a subsequent day in the absence of unequivocal hyperglycemia. Pre-diabetes is the term now used for patients with impaired fasting glucose (IFG) or impaired glucose tolerance (IGT). [8]
Figure 2
An important clinical sign differentiating T1DM from T2DM is insulin deficiency, which most often presents with ketonuria and/or ketoacidosis. This patient was suspicious for T1DM due to his weight loss, metabolic acidosis, and the ketones present in the urine. Although most patients with T1DM are either children or adolescents, the ADA [8] states that age should not be a determining factor in T1DM. Immune mediated diabetes can occur at any age, even in the 8th and 9th decades of life. [8] KP-T2DM and LADA were differential diagnoses for this patient. Diagnostic testing for islet cell antibodies is a definitive way to determine if a patient has autoimmune T1DM versus KP-T2DM. [1112] A pressing concern was that this patient was in metabolic acidosis and could potentially develop DKA. Electrolytes were drawn to evaluate the patient’s acid/base status and determine if there was any worsening in his metabolic acidosis. A lipid profile was ordered since diabetic patients are at higher risk for lipid abnormalities and should be screened. Urine microalbumin was also ordered to evaluate for diabetic nephropathy. [9]
According to the ADA, the four clinical categories of diabetes are 1) Type 1 diabetes mellitus (T1DM); 2) Type 2 diabetes mellitus (T2DM); 3) Other specific types of diabetes, such as single genetic defects in beta-cell function or action, diseases of the exocrine pancreas, or chemical or drug induced diabetes; and 4) Gestational diabetes. T1DM accounts for only 5-10% of the cases of diabetes worldwide. T1DM is further divided into two categories: Type 1-A (Immune-mediated), and Type 1-B (Idiopathic, non-autoimmune). [8] The majority of cases of T1DM are Type 1-A, which is caused by destruction of the beta cells in the islets of Langerhans through either a humoral or cellular autoimmune response. Type 1-A, or immune mediated T1DM is responsible for 90-95% of all cases of the disease. [13] Positive ICAs are a strong indicator of immune mediated T1DM. Antibodies to several of the autoantigens, such as glutamic acid decarboxylase-65 (GAD65), protein tyrosine phosphatase IA-2 (IA-2), and insulin autoantibody (IAA), are highly predictive of T1DM. [14]
Some researchers have suggested additional categories of diabetes such as Latent Autoimmune Diabetes of Adults (LADA) and a nonautoimmune subcategory of Type1-B known as Fulminant Type-1. [11121516] LADA has a more insidious onset, and can usually be managed with oral hypoglycemics initially. Patients with LADA also have a phenotypic presentation more closely resembling T2DM, including obesity and central adiposity. Patients with LADA typically do not require insulin at the time of initial diagnosis. The term LADA was created to describe a subset of adult patients that are initially non-insulin requiring, but have the autoimmune markers of T1DM and go on to require insulin within a few years. [11] The Immunology of Diabetes Society has defined LADA as follows: 1) age over 30; 2) positive for at least one of the four antibodies commonly found in T1DM (ICA, IAA, IA-2, and GAD-65); and 3) not requiring treatment with insulin within the first 6 months after diagnosis. [1617]
Ketosis-prone T2DM is characterized by an acute presentation of ketoacidosis, a strong family history of T2DM, negative autoantibodies, and an intermediate insulin response to meals. In addition, these patients do not require long-term insulin administration to maintain glycemic control. Once euglycemia is achieved with insulin at the initial event, patients can be switched to oral hypoglycemics. [12] Other researchers have labeled this subtype “Flatbush diabetes”, Idiopathic type-1 diabetes, and type 1-b diabetes. These patients are usually obese, African-American or Hispanic, with negative genetic and autoantibody markers. [18] Although these patients may be managed with oral hypoglycemics for some time, they progress to insulin requirement more rapidly than patients with T2DM. [12]
General Therapeutics
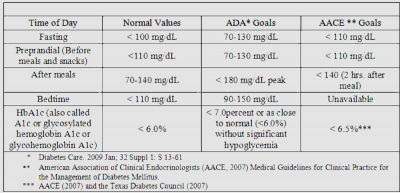
The patient was asked to monitor blood glucose four times a day for the next two weeks: fasting each morning, at bedtime each night, and before and two-hours after one meal each day. He was instructed to alternate which mealtime blood sugar to check each day. In addition, he was asked to test his glucose at 3:00 or 4:00 AM at least two times in the next two weeks. He was instructed to keep a three-day food diary and glucose measurements and bring these to the next appointment. The timing and frequency of self-measurement of blood glucose (SMBG) depends upon the type of diabetes, the needs and goals of the patient and the level of glucose control desired. [9] One of the main reasons for self-glucose measurement is to assist in medication adjustment, diet changes, activity level, or all 3 factors. [19] T1DM patients ideally should monitor their blood sugar before meals, after meals and at bedtime. Since this schedule can mean checking glucose levels 7-8 times a day, a realistic alternative is to have the patient check plasma glucose before every meal, two hours after one (different) meal each day, and at bedtime every day for a limited time to establish where the elevations occur most. This invaluable information allows the practitioner to adjust the patient’s medications and insulin appropriately. Diabetes is a disease that is primarily controlled by the patient. Diet, exercise, and blood glucose monitoring as well as appropriate medication management are important in the control of diabetes. Table 3 lists the American Diabetes Association (ADA) recommendations for blood glucose targets. The American Association of Clinical Endocrinologists (AACE, 2002) has stricter guidelines for blood glucose targets [20] (see table 3 below).
For diabetic patients taking two-to-three insulin injections per day or more, the blood sugar should be monitored three to four times per day, usually before giving an injection and at bedtime. [9] For patients with continued elevation of fasting glucose, or patients who may have low blood glucose reactions during the night, a 3 AM check may also be needed to differentiate between the “dawn phenomenon”, the “Somogyi effect”, and waning bedtime or evening insulin levels. [2919]
Correct differentiation between these phenomenon is important for proper insulin adjustment. Pre-breakfast hyperglycemia is sometimes due to the Somogyi effect in which nocturnal hypoglycemia causes release of counter-regulatory hormones that produce hyperglycemia by 7:00 AM. However, the more common cause of pre-breakfast hyperglycemia in patients with T1DM and the majority of patients with T2DM is the waning of evening or bedtime insulin and/or the “dawn phenomenon”. The “dawn phenomenon” is due to the normal physiologic reduction in insulin sensitivity to insulin between 5 AM and 8 AM as a result of spikes of growth hormone that were released at the onset of sleep. [2] An extremely low blood glucose level at 3 AM indicates that pre-breakfast hyperglycemia is a result of the “Somogyi effect”, while a normal or slightly elevated glucose level is indicative of the “dawn phenomenon”. An elevated 3 AM blood glucose level indicates waning evening or bedtime insulin levels. Table 4 illustrates the differences and recommended insulin adjustments.
Medications
1. Initiate Insulin
Insulin glargine (Lantus ® ) was prescribed and the patient was instructed to take 15 units subcutaneously at bedtime. The patient was given instructions to increase the glargine insulin dose by two units every three days if the three-day average fasting plasma glucose was greater than 120 mg/dL. [A basal/bolus regimen was to be started later, once he had mastered the once daily basal insulin and after the fasting blood glucose levels began to decrease].
The normal pancreas secretes 25-30 units of insulin daily. [2] Patients with a body mass index (BMI) greater than 25kg/m2 should be started on 10-15 units of intermediate insulin (such as NPH) or long-acting glargine at bedtime. [2122] Titration by two units every three to seven days, using a dose titration schedule based on self-monitored FPG, should be initiated with a target of less than 100 mg/dL. [1923] If the patient is known to have insulin deficiency, replacement of both basal and prandial insulin should be initiated as soon as possible using a 50/50 ratio [23] . When instituting a basal-prandial regimen, patients should decrease the calculated 50 percent basal regimen by 20 percent to avoid hypoglycemia. Intermediate insulin has an onset of action of between two to four hours, achieves a peak in approximately six to seven hours, and can last up to 20 hours. [21] Long-acting insulin glargine has an onset of action of two hours, reaches a plateau of action at four to six hours, and lasts up to 24 hours. [2124] In general, every one to two unit increase in insulin leads to a 30-50-mg/dL decrease in glucose concentration. [2122]
2. Discontinue Metformin and Pioglitazone:
The patient was instructed to discontinue metformin and pioglitazone. Metformin may increase the risk of lactic acidosis, especially in patients with metabolic acidosis or impaired renal function. [2526] Pioglitazone was discontinued because the patient was started on insulin. The pioglitazone package insert recommends not using this medication in patients with ketoacidosis, or who are insulin deficient, since the mechanism of action is dependent on the presence of insulin. The package insert also recommended that pioglitazone be taken at least three months before maximum results are seen, therefore this medication would not be able to rapidly bring this patient’s hyperglycemia under control. [126]
3. Begin Aspirin (ASA) 81 mg once daily
Aspirin should be used as a primary or secondary prevention strategy in patients with known cardiovascular disease (CVD), or with T1DM or T2DM at increased risk including those over 40, or with a family history of CVD, who smoke, have HTN or dyslipidemia. [8] The recommended dose for prevention of cardiovascular events is 81mg of ASA. [27]
Education
The patient was given one-on-one counseling and education regarding the pathophysiology of diabetes and the signs, symptoms and actions to take for hypoglycemia, as measured by a blood sugar level of 70mg/dl or less. A prescription for glucagon 1mg kit was given, and his wife was instructed to give him 0.5 to 1mg subcutaneously or intramuscularly if he should become unconscious. The patient was also counseled to use either the glucose tablets or the glucose gel available at the pharmacy or to drink some orange juice or eat a small piece of candy if his blood glucose fell below 70 mg/dl if he became symptomatic; i.e., shaky, diaphoretic, or confused. Gaining an understanding of the earliest symptoms of not only the general symptoms of hypoglycemia such as hunger, perspiration, nervousness, and confusion, but also their individual initial symptoms may help prevent more severe hypoglycemia episodes from occurring, and allows these events to be easily managed with a snack or another glucose source. Educating patients on the differences between major and minor hypoglycemia and how to manage each one is an important part of diabetes education. [28] The patient and his wife were referred for Diabetes Self-Management Education (DSME) with a Certified Diabetes Educator (CDE) for meal planning, carbohydrate counting, and insulin administration education. DSME can help patients obtain tighter glycemic control and achieve more positive outcomes. [29]
Follow-up
The patient was scheduled to follow-up with the Diabetes Specialty Team in 2 days and asked to bring his logs and food diary. Intense education and frequent follow-up would be needed to ensure that he had an adequate understanding of proper insulin administration, carbohydrate counting, dietary changes, his medication regimen, and to ensure that his ketosis and metabolic acidosis were resolving. Adjustments in his medication would continue until his blood glucose levels were controlled and his symptoms alleviated. [8]
Subsequent Appointments
On his second appointment with the Diabetes Specialty Care Team (two days after his initial appointment), the patient stated that he was feeling a little better. Review of his blood glucose logs revealed that AM fasting blood sugars ranged from 170—220 mg/dl. The breakfast two-hour postprandial readings were 180 and 200 mg/dl. His lunch pre-meal glucose readings averaged 150 mg/dl, and evening pre-meal readings were 178 and 180 mg/dl. Two-hour postprandial evening meal readings were between 170-200 mg/dl. He had two – 3:00 AM readings that were 120 mg/dl and 100 mg/dl respectively. He had no symptoms of hypoglycemia and had not experienced any hypoglycemic events. He stated that his energy level had increased, and his vision was less blurry. He had been checking his blood glucose as requested. He denied any chest pain, shortness of breath, edema, or muscle pains or weakness. He had increased his glargine insulin from 15 to 17 units at bedtime.
Subsequent Laboratory Results
Review of laboratory results ordered at the first appointment with the Diabetes Specialty Care Team that revealed that two of the three Islet Cell Autoantibodies (ICAs) tests were positive. Glutamic acid decarboxylase – 65 (GAD-65) and protein tyrosine phosphatase IA-2 (IA-2) were both positive. The other ICA - Insulin Autoantibody, (IAA) was negative. The positive antibodies provided proof of autoimmune diabetes [30] . The patient did not fit the phenotypic characteristics or the diagnostic criteria for LADA, since he required insulin at the time of his diagnosis to correct his ketosis. Additionally, the positive autoantibodies rule out KP-T2DM (see prior discussion). The fasting C-peptide was 2.9 (normal = 0.8-4.0). A normal C-peptide level indicated that he still had functioning beta cells. C-peptide levels are considered a reliable marker of residual beta-cell function, however, according to the findings in the Diabetes Prevention Trial – Type 1 (DPT-1), a normal C-peptide is not unexpected at the onset of T1DM clinical disease. [3031] The Diabetes Control
and Complications Trial (DCCT) also found that a significant amount of beta cells are still functioning and an increased prevalence of residual insulin secretion is more common in those individuals diagnosed with T1DM after the age of 18 years of age. [32] The remainders of the patient’s laboratory results are shown in Table 5. Additionally, the random urine microalbumin was negative, the urinalysis was now negative for ketones and urine glucose had decreased from 4+ to 2+.
Long-Term Follow-up
The patient was seen again 1 week and 2 weeks after the initial office appointment with the diabetes specialty care team and continued to do well. The glargine insulin was titrated up over the following 3 weeks to 25 units each night at bedtime. He was also started on rapid-acting aspart insulin (NovoLog ® ) at mealtimes. This rapid-acting insulin should be injected 5-15 minutes prior to mealtimes. [21] Intensive insulin therapy is initiated in a 1:1 basal:bolus regimen based on total daily insulin (TDI) dose of 0.3-0.5 units/kg/day. Fifty percent of this TDI dose is given as the long acting basal insulin (glargine) and the remaining fifty percent given as rapid-acting (RAI) insulin (aspart). [1933] His fasting blood sugars significantly improved and now ranged from 90-120 mg/dL with no hypoglycemic episodes. The patient quickly mastered carbohydrate counting, and was started on a 1:15 insulin to carbohydrate ratio for his bolus insulin. The insulin: carbohydrate ratio is used to calculate the number of grams of carbohydrates that will be covered by 1 unit of rapid acting insulin (RAI). To determine this ratio, divide 500 by the TDI dose (1 unit RAI covers 500/TDI). [1933] The patient’s total daily insulin dose was 40 - 43 units, or about 0.36 units/kg body weight. Addition of a correction factor for his bolus insulin may be necessary in the future. HbA1c, lipids and complete metabolic profile (CMP) will be drawn every three months.
Based on lipid results, this patient also had primary mixed hyperlipidemia (type 5) which is characterized by the pathologic presence of chylomicrons after a 12-14 hour period of fasting. [34] People with central adiposity, diabetes, and the metabolic syndrome often have elevated triglycerides and low HDL. [35] Some authorities use the total cholesterol to HDL ratio as an indicator of lipid-related coronary risk. A lower ratio means a lower risk for a coronary event. However this patient’s ratio is 183/25 – or a 7.32 times higher risk for coronary heart disease (CHD). [36] The lipid profile of patients with T1DM is highly dependent on glycemic control. Individuals with poorly controlled T1DM frequently have high levels of triglycerides and LDL-cholesterol, but levels usually improve with improved glycemic control. [37]
The patient was started on extended release niacin (Niaspan ® ) 500 mg once daily (to be titrated to 2000 mg daily by increasing the dose by 500 mg every 2 -3 weeks as tolerated) to improve the lipid profile. If HDL is less than 40 mg/dL and LDL between 100-129 mg/dL, addition of a fibric acid derivative or niacin is advised. A modest dose of niacin (750-2,000 mg/day) significantly benefits LDL, HDL, and triglycerides. [93438] In the NAUTILUS study by Vogt et al., HDL was increased by 24 percent and triglycerides were reduced by 13 percent at week 15 in patients reaching the target goal of 2000 mg/day. [39] The PROCAM study found that increased triglycerides were an independent risk factor for cardiovascular disease. [40] This study found that daily consumption of up to three grams of niacin could lower plasma triglycerides by up to 45 percent, raise HDL by up to 25 percent, and lower LDL by up to 20 percent. Starting therapy at low doses, and gradually titrating the dose, can minimize the side effects of flushing, dizziness, or itching. Giving the dose with aspirin, or the use of longer-acting preparations, such as Niaspan ® , also reduces side effects. [35] Goals for lipid management are: 1) LDL less than 100 mg/dL [less than 70 mg/dL for patients with diabetes and coronary artery disease (CAD)]; 2) HDL greater than 40 mg/dL in men and greater than 50 mg/dL in women; and 3) triglycerides less than 150 mg/dL. [203438]
Summary
This case represents the difficulty that clinicians may encounter in differentiating between T1DM, T2DM, and other less common types of diabetes mellitus. This patient was initially diagnosed with T2DM based largely on age and phenotype, despite a 40-pound weight loss. A simple dipstick urinalysis revealed large amounts of ketones, which indicated insulin deficiency. The presence of ICAs confirmed the diagnosis of T1DM. Since this patient presented with ketosis requiring insulin within 6 months of diagnosis, he did not fit the criteria for LADA. [1617] This patient also had positive insulin autoantibodies, so he did not fit the criteria for ketosis-prone T2DM. [12]
Prompt establishment of the correct diagnosis in this case was critical in preventing further deterioration of his condition and development of diabetic ketoacidosis. Of notable significance was the fact that a thorough history revealing the patient’s recent weight loss was the single-most important factor that triggered the APN to proceed with further work-up.
Results of the Diabetes Control and Complications Trial (DCCT) clearly show that achieving near-normal glycemia in patients with diabetes reduces the risk for long-term microvascular and neurological complications such as retinopathy, nephropathy and neuropathy. [32] With early intervention to regain glycemia, this patient will have a reduced risk for developing the long-term complications of diabetes. According to the findings in the DCCT trial, each 1% decrease in the HbA1c (e.g., from 8% to 7%) decreases the risk of macrovascular complications by 40%. [32] Addressing both macrovascular and microvascular risk factors by controlling blood pressure, lipids and cardiovascular risk factors is also extremely important. [9] While intensive insulin regimens aimed at lowering blood glucose to near normal levels has been shown to reduce microvascular risks, the risk for serious hypoglycemia remains. Continued education and encouragement of patient involvement in the control of their diabetes is essential for a positive prognosis. Diabetes is a chronic, lifelong illness that requires management of the disease by the patient. Numerous sources of excellent patient information and education as well as clinician support exist (see Table 6).
Continued support, education, and collaboration between the health care team, the patient and his family will offer the best alternatives for positive outcomes (see Table 7).
Acknowledgements
This author would like to thank the following individuals for their support and assistance in the editing of this manuscript: Pamela Willson, PhD, RN, FNP-BC, Terry Dickey, MSN, RN, FNP/GNP-BC, and Amber Jones, ME. In addition, this author would like to extend many heartfelt thanks to Joanne Hickey, PhD, RN, ACNP-BC, FCCM, FAAN for her continuous support and motivation, and to Susan Ruppert, PhD, RN, ANP-BC, NP-C, FCCM, FAANP for her continued encouragement during the writing of this manuscript.
Correspondence to
Elaine Ballard, MSN, RN, FNP-BC 1910 Cross Point Rd, McKinney, Texas 75070 972-529-5481 email: elaine@ballard.name